In my years spent behind the bench, I’ve realized that the difference between a "good" chromatogram and a "publishable" one usually isn't the instrument—it's the sample prep. If you are struggling with messy baselines, ghost peaks, or poor recovery, you need to master solid phase chromatography and its most practical application: solid-phase extraction (SPE).
Understanding the Fundamentals: What is Chromatography?
If someone asks what is chromatography, the standard textbook answer involves "the separation of mixtures." But to a practitioner, chromatography what is actually translates to control. It is the art of using physical chemistry to force different molecules to move at different speeds.
Whether you are performing complex HPLC or simple TLC, the question of how does chromatography work boils down to a fundamental tug-of-war. To understand what chromatography is, you must look at the two competing environments:
-
The Stationary Phase: This is the "anchor." It stays put inside your column or on your plate. What is the stationary phase in chromatography? In most labs, this is a solid adsorbent, like silica or a bonded phase (like C18), providing the surface area for your analytes to interact with.
-
The Mobile Phase: This is the "carrier." It is the liquid or gas that pushes your sample through the system. What is the mobile phase in chromatography? This is the solvent (in liquid chromatography) or carrier gas (in gas chromatography) that must be carefully chosen to balance its affinity for the analyte against the stationary phase's grip.
.png)
Fundamental Chromatography Process
When I explain chromatography in chemistry to new lab technicians, I tell them to imagine a crowded hallway (the stationary phase in chromatography) and a gust of wind (the mobile phase in chromatography) blowing leaves through it. Some leaves stick to the walls more often; others fly straight through. That difference in "stickiness" is what gives us our separation. The magic happens in the specific interaction between the mobile phase and stationary phase in chromatography.
Solid-Phase Extraction (SPE): The Secret to Clean Samples
Now, let’s move from the "analytical" side to the "preparative" side. What is solid phase extraction? Solid-phase extraction (SPE) is essentially chromatography's more rugged, faster cousin. While analytical chromatography aims to separate every component to see it on a detector, SPE is designed to "clean up" or concentrate your sample before it ever touches your expensive analytical column. In our lab, we use SPE to isolate analytes from messy matrices like blood, urine, or environmental water.
.png)
Steps of Solid-Phase Extraction (SPE)
Chromatography: How Does It Work in SPE?
Instead of a long, high-resolution column, we use a small plastic cartridge. The process follows a logical flow:
-
Conditioning: Wetting the sorbent to prepare the surface.
-
Loading: The sample passes through. The analytes of interest "stick" to the mobile phase stationary phase chromatography interface, while the junk flows through.
-
Washing: Removing impurities without dislodging the target.
-
Elution: Using a "strong" solvent to pull the purified analyte into a collection tube.
Analytical Precision: How Does Liquid Chromatography Work?
When we scale this up, we look at how does liquid chromatography work. Here, we use high pressure to force the interaction to happen faster and with higher precision. In a modern LC system, how does liquid chromatography work involves a high-pressure pump, an injector, and a column packed with very small, uniform particles. This allows us to separate compounds that are chemically very similar—something a simple SPE cartridge can't always do.
Strategic Selection: Normal Phase vs. Reverse Phase
The most important decision you’ll make is the polarity of your system. This brings us to the classic laboratory debate: normal phase vs reverse phase.
1. Normal Phase HPLC
In the early days, the "normal" way involved a polar stationary phase (like bare silica) and a non-polar mobile phase (like hexane). This is normal phase hplc. In this setup, "like attracts like"—polar analytes stick to the polar silica. I usually choose normal phase chromatography vs reverse phase when dealing with very hydrophobic samples, such as oils or isomers.
2. Reverse Phase Chromatography
Today, 90% of our work uses reverse phase vs normal phase. In reverse phase chromatography vs normal phase chromatography, we flip the script. The stationary phase is non-polar (usually C18), and the mobile phase is polar (water/acetonitrile).
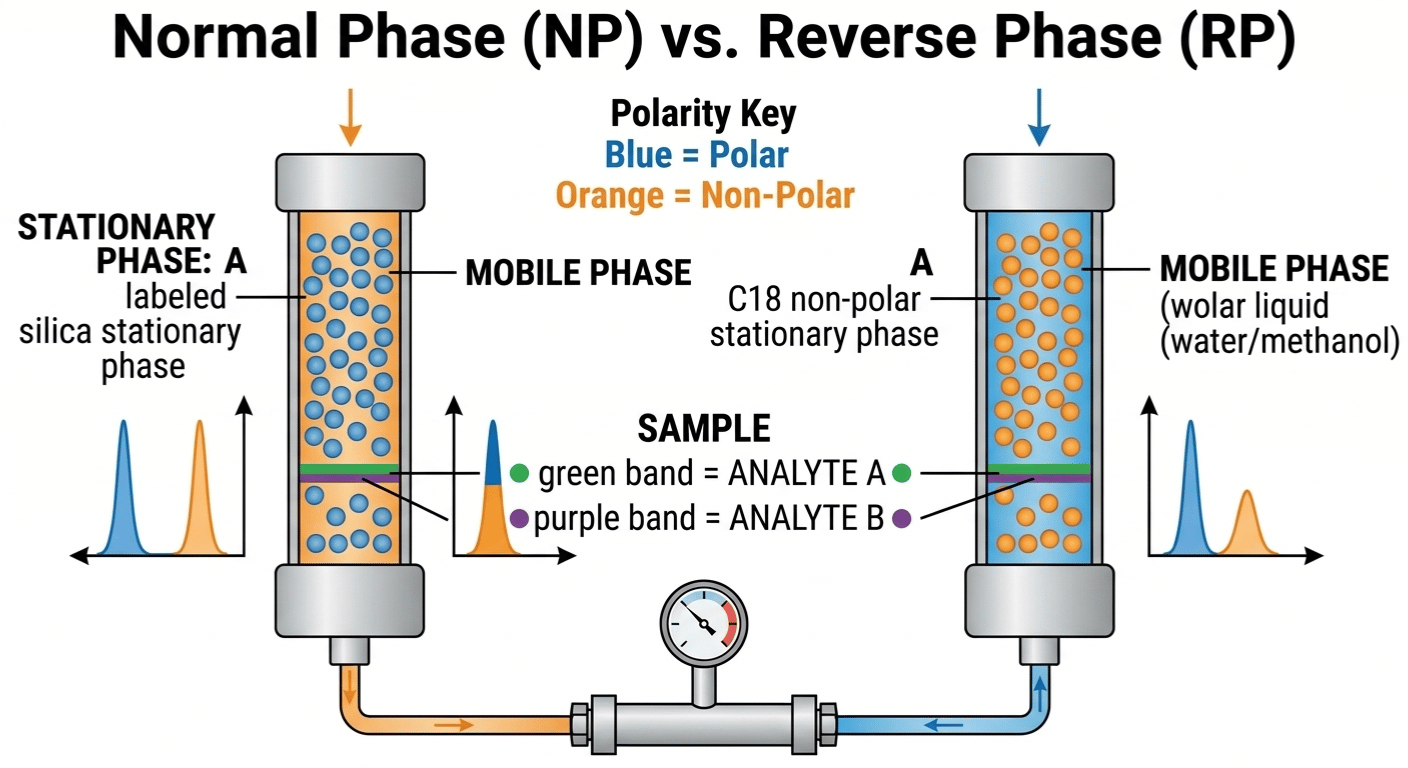
Comparison: Normal Phase vs. Reverse Phase
In a reverse phase vs normal phase hplc comparison, reverse phase is the winner for robustness. Most molecules we care about—pharmaceuticals and metabolites—are water-soluble, making reverse phase vs normal phase the logical choice for modern labs.
Comparison Table: Normal Phase vs. Reverse Phase HPLC
-interaction.png)
Molecular View: Reverse Phase (C18) Interaction
| Feature |
Normal Phase HPLC |
Reverse Phase HPLC |
| Stationary Phase |
Polar (Silica, Alumina) |
Non-polar (C18, C8, Phenyl) |
| Mobile Phase |
Non-polar (Hexane, Heptane) |
Polar (Water, Acetonitrile) |
| Analyte Polarity |
Best for very polar samples |
Best for mid-to-low polarity |
| Elution Order |
Least polar elutes first |
Most polar elutes first |
Expert Recommendations for Your Lab
-
Equilibration: Whether you are using solid phase chromatography or SPE, always ensure your stationary phase is properly wetted.
-
pH Control: For ionizable compounds, pH can completely change your results. A small shift can make a molecule go from "sticking" to "flying through."
-
Efficiency: Don't waste time with manual liquid-liquid extraction when SPE can provide higher purity in half the time.
Need Expert Advice on Your Sample Prep?
Choosing the right sorbent or the perfect mobile phase shouldn't be guesswork. Whether you are setting up a new SPE protocol or troubleshooting a stubborn HPLC method, our team is here to help you get the recovery rates you deserve.
Contact us for a technical consultation or a quote on high-purity consumables:
References & Technical Sources
-
USP General Chapter <621>: "Chromatography" – The industry gold standard for chromatographic separation and system suitability.
-
Buszewski, B., & Szultka, M. (2012): "Past, Present, and Future of Solid Phase Extraction: A Review." Critical Reviews in Analytical Chemistry.
-
Hennion, M. C. (1999): "Solid-phase extraction: method development, sorbents, and coupling with liquid chromatography." Journal of Chromatography A.
-
Anastassiades, M., et al. (2003): "Fast and Easy Multiresidue Method (QuEChERS) for Determination of Pesticide Residues in Produce." Journal of AOAC International. (Focus: Modern SPE applications in food safety).
-
U.S. EPA Method 525.2: "Determination of Organic Compounds in Drinking Water by Liquid-Solid Extraction and Capillary Column Gas Chromatography/Mass Spectrometry."


 English
English
 Chinese
Chinese
.png)
.png)

-interaction.png)
