Mar. 13th, 2026
Introduction
I’ve been where you are—staring at a monitor at 2 AM, watching a baseline drift like a lost boat at sea, wondering if the "HPLC doo-hickey" was invented just to test my sanity. If you're feeling delirious trying to wrap your head around high performance liquid chromatography (HPLC), take a breath. It’s not just a box of expensive parts; it’s a delicate dance between chemistry and physics.
In the lab, we often say the mobile phase is the "life line" of the system. Without it, your sample is just sitting in an injector, going nowhere. But how do you choose the right "train" to carry your molecules to the detector? Let’s break it down like we’re chatting over a coffee in the breakroom.
1. What is HPLC, Really?
At its core, HPLC is about separation. Think of it like a race where the "track" is the stationary phase (the column) and the "wind" pushing the runners along is the hplc mobile phase.
The magic happens because different molecules have different "affinities." Some molecules love the mobile phase and zoom through the column. Others are "sticky"—they have a crush on the stationary phase and lag behind. This difference in speed is what creates those peaks on your chromatogram.
2. Normal Phase vs. Reverse Phase
This is usually where everyone starts pulling their hair out. In my years of practice, I’ve found the easiest way to remember this is by looking at polarity.
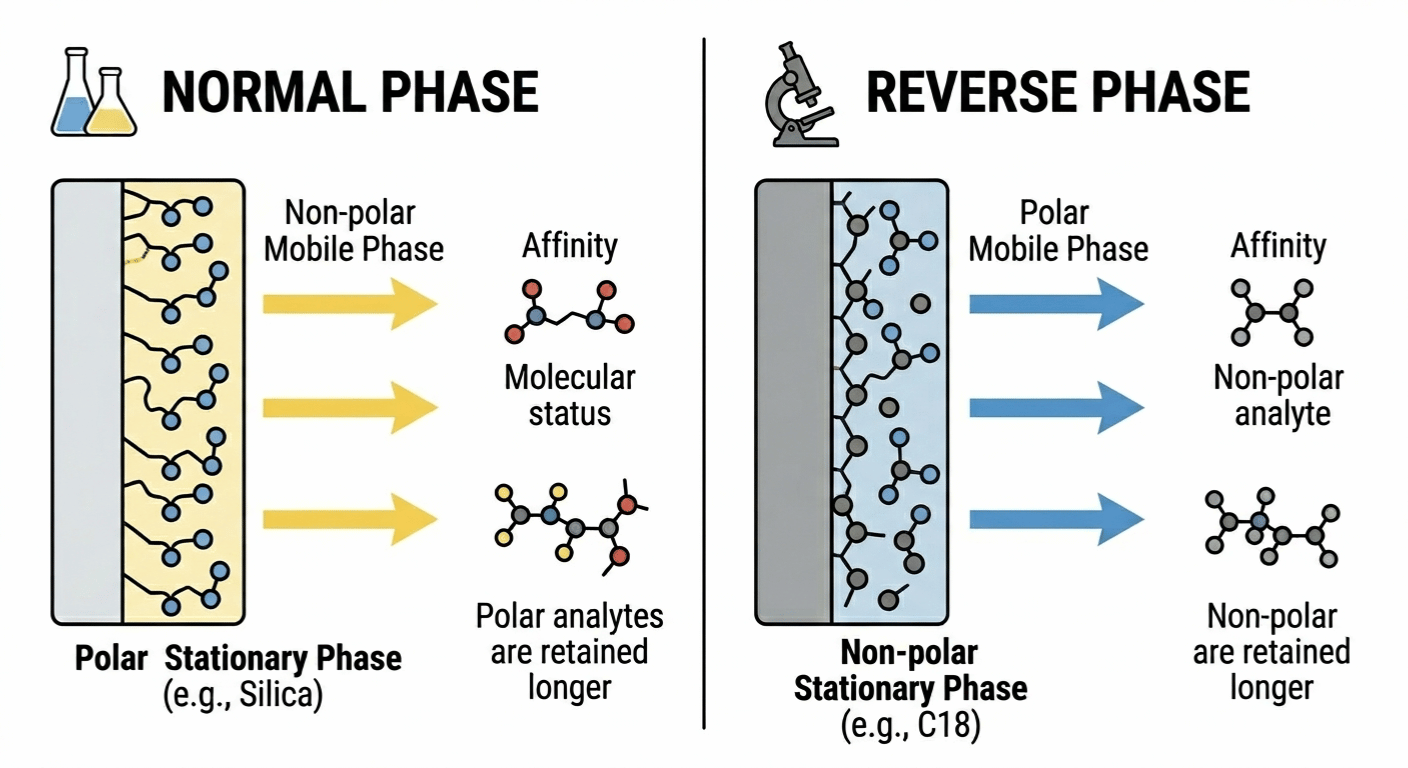
A. Normal Phase Chromatography
This is the "old school" way. In normal phase chromatography, the stationary phase is polar—think of it like a magnet for polar molecules. The mobile phase we use here is non-polar, usually solvents like hexane or heptane.
-
The Logic: Polar "sticky" molecules get held back by the polar column. Non-polar molecules wash out quickly with the non-polar mobile phase.
-
The Pro Tip: If you increase the polarity of your mobile phase (adding a bit of isopropanol), your samples will actually move faster.
B. Reverse Phase Chromatography
Today, this is the gold standard. Probably 90% of the work I do in the lab involves reverse phase chromatography. Here, we flip the script. The column is non-polar (hydrophobic, like oil), and the phase mobile is polar (usually a mix of water and methanol or acetonitrile).
-
The Logic: Water-loving (polar) molecules zoom through because they hate the oily column. Fat-loving (non-polar) molecules stick to the column like glue.
-
Why we love it: It’s predictable, versatile, and uses water-based solvents which are easier to manage.
3. The "Golden Rule" of Solvent Quality
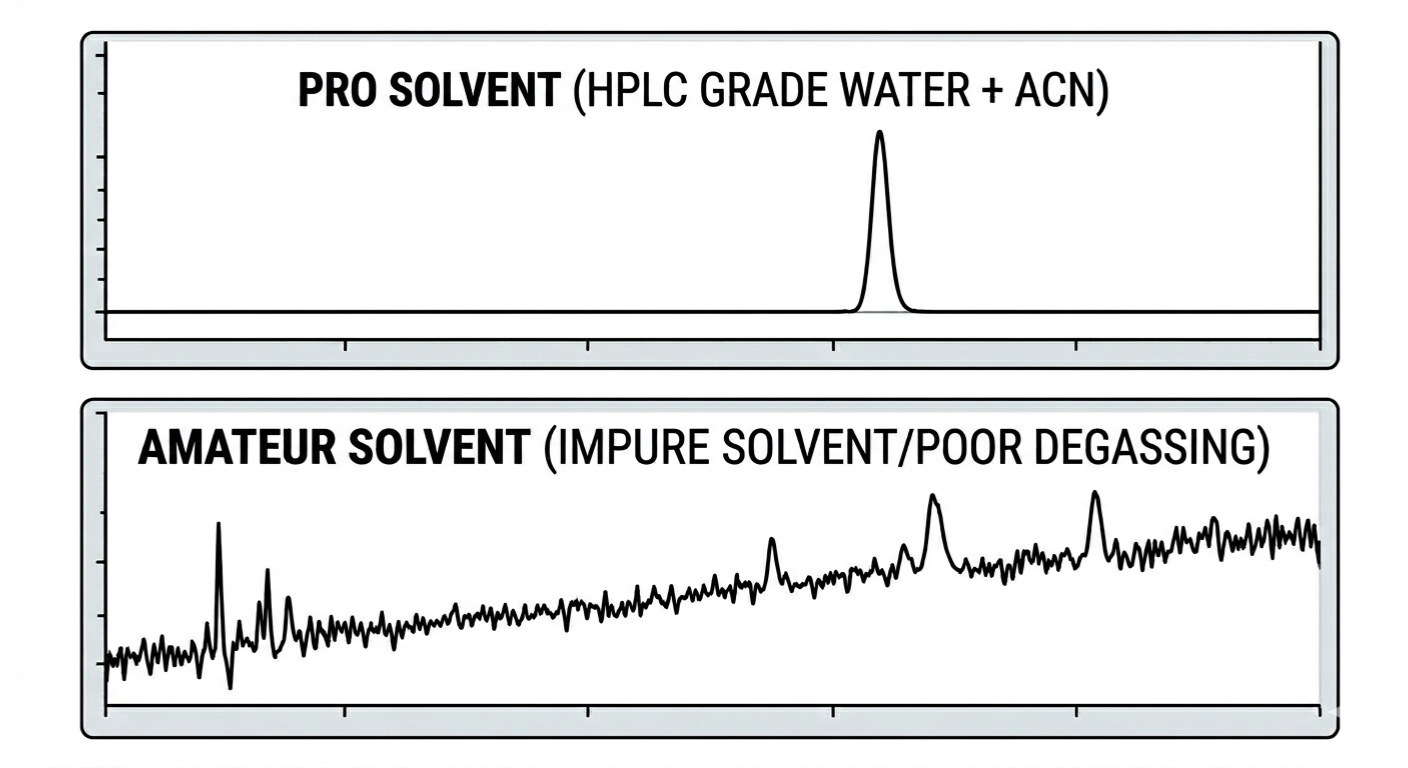
You’ll see "HPLC Grade" on labels for a reason. Don't even think about using ACS grade or anything less. Why? Because those "tiny" impurities in lower-grade solvents will accumulate on your column over time, killing your resolution and creating weird ghost peaks.
Always use Milli-Q (18.2 MΩ.cm) grade water. If your water is "dirty," your baseline will look like a mountain range, and you’ll spend three days trying to figure out why.
4. Degassing and Filtration: The Performance Killers
Air is the enemy of the hplc mobile phase. Under normal pressure, air dissolves in your solvents. But once that solvent hits the pump, those tiny bubbles can get trapped in the check valves or the detector cell.
-
The Result: Flow rate fluctuations, pulsing baselines, and moving retention times.
-
The Fix: Use built-in degassers, or sonication/vacuum filtration if you're running high-sensitivity assays.
Filtration is Not Optional. Even HPLC grade solvents can have microscopic particles from the manufacturing process or the bottle cap. Always filter through a 0.22µm or 0.45µm membrane before it touches your reservoir. It’s a 5-minute task that can save your column from a premature death.
5. Buffers and the Glass Trap
In reverse phase chromatography, we use buffers to keep pH stable for ionizable samples.
-
Pro Tip: If you used a buffer today, wash the system with pure water/organic mix before you go home. If you don't, those salts will crystallize and shred your pump seals.
-
The Glass Trap: Did you know that if your hplc mobile phase pH is > 8.0, it can actually leach metal ions from glass reservoirs? Switch to stainless steel or specialized plastic containers in these cases.
Let’s Get Your Separation Perfect
Getting the mobile phase right is 80% of the battle in HPLC. If you’re struggling with "shag carpet" baselines, pressure spikes, or inconsistent results, don't waste another day guessing.
I’ve spent thousands of hours at the bench troubleshooting these exact issues. I’m always happy to share a bit of "lab-proven" advice to get your project back on track.
References
-
Snyder, L. R., Kirkland, J. J., & Dolan, J. W. (2011). Introduction to Modern Liquid Chromatography. 3rd Edition. John Wiley & Sons. (The "Gold Standard" for understanding HPLC separation mechanisms).
-
Dolan, J. W. (2002). "Mobile Phase Preparation." LCGC North America, Troubleshooting Series. (Essential practical guide for degassing and filtration).
-
Aparicio, R. (Ed.). (2013). Manual of HPLC Optimization. Elsevier. (Comprehensive data on solvent polarity and buffer selection in reverse phase chromatography).
-
Agilent Technologies. "HPLC System Troubleshooting Guide: Avoiding Common Mobile Phase Errors." Technical Resource Library.
-
Waters Corporation. "Principles of Chromatography: Normal Phase vs. Reverse Phase." Education Series.
-
International Conference on Harmonisation (ICH). Q2(R1) Validation of Analytical Procedures: Text and Methodology. (Standard for peak area quantification and retention time stability).


 English
English
 Chinese
Chinese